An effortless and comprehensive approach to burden of illness reviews
Blog - HEORO PRO INSTRUMENT ONTOLOGY HAS OVER 4,200 ITEMS
Published: 30-05-2016
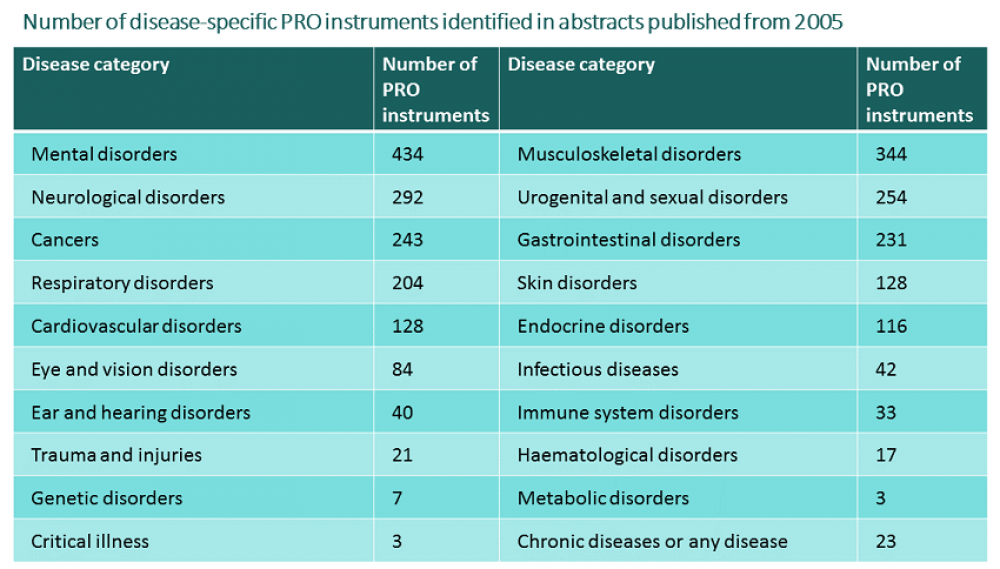
The new heoro.com database launched successfully at ISPOR DC last week. We've developed an ontology of the 4,250 PRO tools we identified from more than 22,200 PRO study abstracts in the database. Creating a new ontology is time-consuming work. We based the disease ontology in heoro.com on MeSH terms from PubMed, the source of the 100,000-plus abstracts we identified from a systematic search from 2005. Our intervention ontology also draws on MeSH terms for treatments, combined with lists of pharmaceutical products licensed in a number of countries including the US and UK, and is updated as studies on new products are published. With our patient-reported outcome (PRO) instrument ontology, though, we started from scratch: First, we identified all the instruments measuring quality of life, utilities, and other patient, family, caregiver or clinician outcomes, experiences and views from the 3,915 abstracts we identified in our training set of 10,000 abstracts moderated by experts in clinical medicine and health economics and outcomes research (HEOR). Then, we developed unique natural language processing software, with around 95% accuracy for identifying PRO study publications in the 90,000 unmoderated papers in the database. The software identified a further 18,339 abstracts that were likely to be PRO studies. Finally, our experts supplemented and indexed the list of PRO instruments by manually going through all 18,339 abstracts. The tables below summarise what we found. 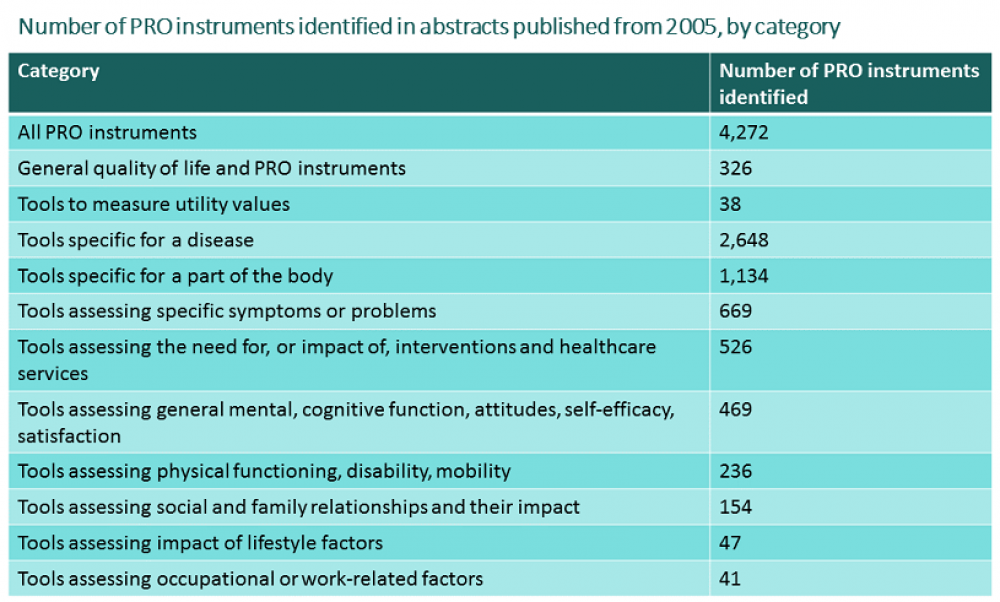
 New abstracts that are imported into the heoro.com database every week are indexed to the relevant PRO tools cited in the abstract, and new instruments are added to the ontology as the abstracts are moderated by our experts. This means that users of heoro.com can rapidly find publications that have used a particular PRO instrument, can find utility values for a specific geographical setting in the general population or associated with a specific disease or intervention, or can identify studies that validated PRO instruments or mapped them to utility values for a particular disease. See more about heoro.com here, or click here to access the database.
New abstracts that are imported into the heoro.com database every week are indexed to the relevant PRO tools cited in the abstract, and new instruments are added to the ontology as the abstracts are moderated by our experts. This means that users of heoro.com can rapidly find publications that have used a particular PRO instrument, can find utility values for a specific geographical setting in the general population or associated with a specific disease or intervention, or can identify studies that validated PRO instruments or mapped them to utility values for a particular disease. See more about heoro.com here, or click here to access the database.